When the slam of a door chopped off the tip of her pinky, doctors told Deepa Kulkarni nothing could be done. She did her own research and proved them wrong.
STORY HIGHLIGHTS
- Sometimes doctors don't know about new treatments being used by other physicians
- Don't hesitate to e-mail a doctor who's a leader in the field that could help you
- That treatment that looks perfect on the internet might work for you -- but it might not
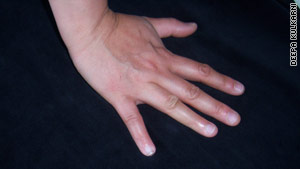
(CNN) -- After running inside from a rainstorm one Friday evening last January, Deepa Kulkarni leaned against the doorway with her right hand to take off her boots. Then, in an effort to make sure the dog didn't get out, someone slammed the door hard, and it landed right on her pinky.
Kulkarni thought the door had only bruised her finger, but then she looked down and saw the tip of her pinky lying on the floor.
"I swooped down and picked it up before the dog got it," she remembers. "At first I was fine, but then I saw the blood -- there was so much blood -- and I felt woozy."
Her husband, Ajit, called an ambulance, and as soon as his parents arrived to take care of their two young children, Kulkarni retrieved her pinky tip from the freezer -- it had broken off just above the base of her fingernail -- and they headed from their home in Davis, California, to the emergency room.
 Video: Woman grows back finger tip
Video: Woman grows back finger tipThat trip to the hospital was the beginning of a five-week odyssey -- one that involved defying doctors, e-mailing specialists around the country and -- in her words -- being pushy and demanding in order to make her pinky whole again.
'100 percent chance of failure'
"The doctor who was on call at the emergency room told me there was no way he could reattach my pinky," she says. "I didn't like that, so I asked to see a specialist."
An orthopedic surgeon concurred with the ER doctor, and made an additional recommendation: He'd have to amputate even more of the finger so it would heal properly.
"I was like, no way, with so much technology out there, there must be some other way to do this," she said. "But he said he wouldn't even attempt to reattach it. He said was there was a 100 percent chance of failure," she remembers.
Disappointed but defiant, Kulkarni declined the surgeon's treatment, and the doctors sent her home with a bandaged finger and painkillers.
'I am writing to you out of desperation.'
Deepa Kulkarni displays her hand after the accident.
Arriving home at midnight, Kulkarni fell asleep, and the next morning immediately got on her computer. With her nine functioning fingers she searched for hope -- any hope -- that her pinky could be repaired.
Kulkarni, a former human resources manager who is now in law school full time, was accustomed to doing research, but after searching for hours, she still couldn't find any new ideas for reattachment.
Kulkarni's pinky grew back after she persuaded a doctor to administer a procedure she had read about on the internet.
She did, however, find something else: stories on "60 Minutes" and "Oprah" about a relatively new procedure called tissue regeneration, which had made amputated finger parts grow back for other patients.
She went to the website for Dr. Stephen Badylak, the University of Pittsburgh physician who helped pioneer the procedure. A little more searching revealed his e-mail address.
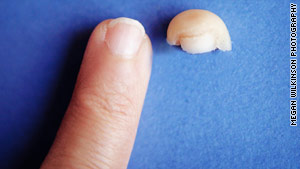
Kulkarni displays her new pinky next to the tip of her old pinky.
"Dear Dr. Badylak," she began. "I am writing to you out of desperation...I ran across your article on the internet and decided to write to you. Can you tell me if [tissue regeneration] works and what my options are to get the treatment?"
She heard back the next day, a Sunday, from one of Dr. Badylak's colleagues. He gave her more details about the procedure, and some bad news: He didn't know of any doctors near her using regenerative medicine.
She e-mailed him photos of her damaged pinky, and he said the team of doctors in Pittsburgh would review them and decide whether they could help her. In the meantime, Kulkarni sent out an all-call in search of a physician near them who used tissue regeneration.
"I e-mailed people at UCLA and Berkeley, and my husband and I called everyone we knew who was a doctor or might know a doctor," she says.
Still, no one had any ideas for her. One friend introduced her to Dr. Malathi Srinivasan, an internist at the University of California-Davis Medical Center, who called Kulkarni from her cell phone while driving home from a skiing trip at Lake Tahoe.
Srinivasan didn't know any doctors with experience in tissue regeneration, but she did give her something Kulkarni badly needed at this point: hope.
"I encouraged her to keep looking, and I did some PubMed searches myself," Srinivasan says, referring to the National Library of Medicine's online collection of research materials.
Seven weeks later, a new pinky
Eventually, Kulkarni made an appointment with Dr. Michael Peterson, an orthopedic surgeon in Davis. At first, Kulkarni says he was hesitant to try tissue regeneration since he hadn't done it before, but she gave him some research materials, and she says eventually he agreed to try it.
The therapy involved cleaning out the finger and removing scar tissue -- a process called debridement -- and then dipping her finger into MatriStem wound powder. After seven weeks of treatment, her fingertip grew back (as shown in the before and after photos above).
"Even now it's not perfect. It's shorter than the other pinky, but just by looking at it you can't tell it was an amputated finger," she says. "I'm able to do everything I could do before. I wash dishes. I cook," she says, adding that she's had physical therapy to decrease tingling in her finger caused by severed nerves.
Kulkarni still keeps her severed pinky tip in the freezer. She thawed it out so CNN could take a photograph of the pinky tip alongside her regenerated finger.
Five lessons learned
Here are lessons Kulkarni learned through her pinky ordeal, plus a few from the doctors who worked with her:
1. Realize your doctor doesn't know everything
"I grew up in India, and doctors there were looked up to as being next to God, and they knew everything and could cure you," Kulkarni says. "What I found out is that even though I like my doctors, I don't have to take every recommendation they give me. I can do my own research."
Srinivasan, the physician who encouraged Kulkarni not to give up, says sometimes doctors don't know about new, cutting-edge treatments being used by other physicians.
"Some doctors are more out-of-date or up-to-date than others," says Srinivasan, an associate professor of medicine at UC-Davis Medical Center who does research on shared decision-making between doctors and patients.
Kulkarni said even when her prospects looked dim, she never completely gave up hope.
"My husband and I are quite pushy, and my husband is smart -- he has a Ph.D. in electrical engineering. We weren't going to take no for an answer," she says.
Petersen, Kulkarni's surgeon, says he's learned from many patients who've come to him having done research on the internet.
"I get a lot of good ideas from patients," he says. "One of the things we have to do as physicians is to listen to our patients."
2. E-mail doctors you don't know
Even though you're a stranger, if you write to a doctor who's a leader in the field, you might get an e-mail in return.
"The doctor from Pittsburgh wrote me back the very next day, on a Sunday," Kulkarni says. "I was so surprised. Maybe I sounded really desperate."
Even if you can't afford to fly out to the expert for treatment, he or she might be able to lead you to a local doctor who can help.
3. Don't be afraid to be a 'bad' patient
Kulkarni said she knows the doctors she saw in the emergency room might have perceived her as an annoying patient when she rejected their treatment options.
"I didn't care," she says. "Imagine if I hadn't pursued other options because I was worried about what other people think -- I didn't want to live with that regret."
4. Be organized
Be fastidious about keeping records. Kulkarni, for example, photographed her pinky at various stages, and she e-mailed the images to various doctors and later to the insurance company to convince them to reimburse her the $1,665 she spent on the drug that regenerated her pinky.
5. Recognize you might be wrong
That treatment that looks perfect on the internet might work for you -- but it might not.
"Sometimes patients are pretty certain through their own research what their problem is and what they need," says Petersen. "It's not up to us as doctors to agree with them. Sometimes part of my role is to guide them that, in reality, that isn't your problem and I don't recommend that treatment."
Learn more about Elizabeth Cohen's book on being an Empowered Patient. 
CNN's Sabriya Rice contributed to this report.